I attended the New England Hemophilia Association’s Consumer Medical Symposium Saturday in Portland, Maine. It was fabulous! Over 200 consumers, medical staff and guest speakers attended. As always, after 31 years in this community, it’s like a family reunion. But it’s also a time of great learning.

We were fortunate to have Dr. Steven Pipe, chair of NHF’s Medical and Scientific Advisory Committee. He provided an excellent and understandable overview of current products, new products and gene therapy, and how they all coexist. It was truly a brilliant presentation. Dr. Pipe started by saying that our community is having some of the most monumental changes in hemophilia that we’ve ever gone through. He talked about standard factor, the factor used prior to newer therapies like extended half-life (EHL) products. Why was EHL developed? To help preserve joints better; to reduce the “burden” of prophylaxis (frequent infusions, wear and tear on the veins). But he reminds us that even though EHL products extend the half life, these products also have a trough, when levels will dip and when a child will be less protected from a bleed.
In discussing the newest therapy, Dr. Pipe calls Hemlibra “substitution therapy.” It is not a factor product. It bridges factor IXa and factor X, bringing them together to allow a clot to form. He made everyone laugh when he called Hemlibra the “eHarmony” of clotting. Factor VIII normally acts as a scaffolding, allowing FIX and FIXa to come together. When there is no factor VIII, the scaffolding cannot support the two. So Hemlibra can now do this. And this new structure looks nothing like factor VIII so it is less likely to encourage inhibitor formation. It has a long half life, in some cases up to 30 days. And it’s administered subcutaneously, so no more trying to secure little wiggly veins. The outcomes of the HAVEN studies were displayed and discussed, causing Dr. Pipe to say that he had never seen anything like them. This nonfactor therapy provides a steady trough as well, not the dipping troughs of EHL or standard factor. It’s also effective in inhibitor and noninhibitor patients. (Download PEN’s Hemlibra article here)
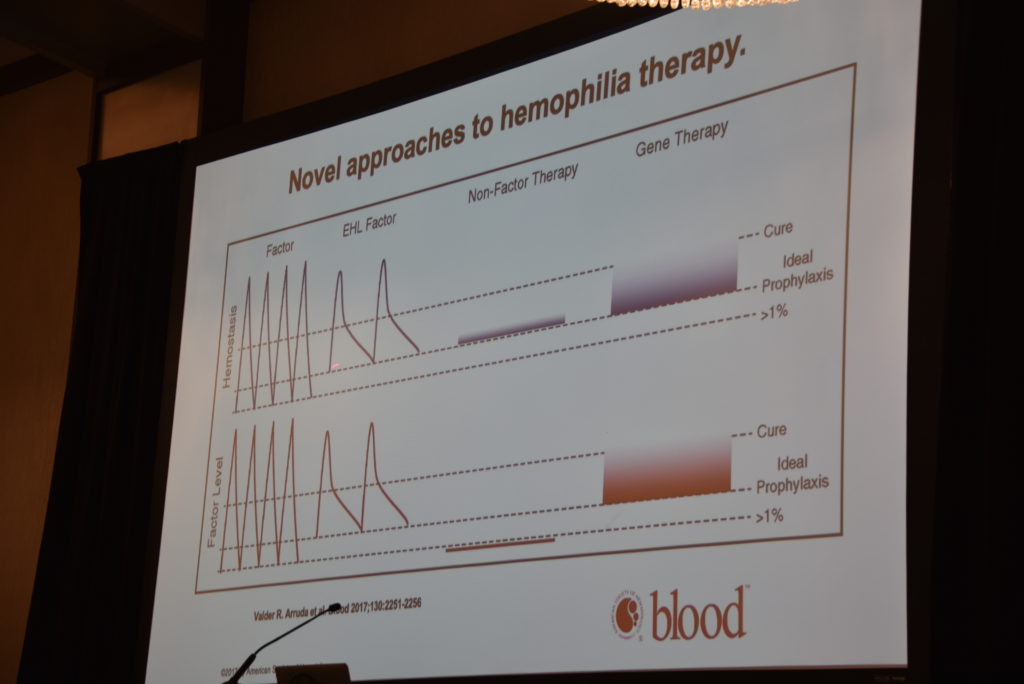
What’s not to like? Well, there are unknowns. Although Dr. Pipe has used Hemlibra in a two year old, patients are being monitored for thrombotic risks, assay issues, annual expenses, and peak bleeding risk events. You can’t do an assay of Hemlibra, unlike factor concentrates. And one third of patients using it will experience some bleeding issues.
Dr. Pipe also showed on a slide the scales of hemostasis; how everything needs to be in balance. Not only do we need to try to clot the blood, but we need the blood not to overclot. The body has its own natural mechnisms for stopping the blood clotting process with naturally occurring antifibrinolytics. Some new therapies will try to remove the antifibrinolytics, which might allow the blood to continue to clot. These are all low burden therapies, desgined to restore the balance in hemostasis.
Dr. Pipe used Amazon.com as a metaphor for how gene therapy could work. The box your order comes in is like the vector carrying the human gene that will be inserted into the body. It’s targeted to the liver, and once implanted, will manufacture factor VIII normally. There are currently 5 gene therapy trials underway for FVIII and even more for FIX.

And I learned something new. Victoria Kuhn asked a pertinent question: why weren’t women being included in the clinical trials of these products and gene therapy? Dr. Pipe replied that women and children were considered “unique high risk groups” by the ethics committee of the FDA. A woman’s eggs could be affected by gene therapy; no one knows for sure. The good news is that Dr. Pipe believes gene therapy will be viable in 2020 or 2021.
He ended his superb presentation by agreeing that there is no one product that everyone should be on, as everyone’s body is different. Glenn Roy, a nurse from Maine, agreed and added that his center does PK studies on every patient, to determine the best therapy for them. What’s important to you? Shaving off one extra infusion? Avoided vein wear and tear? Out of pocket costs?
Mary Fitzgerald, a mother of an adult son with hemophilia who I have known for decades, reminded the younger parents in the audience to be cautious about rushing to new therapies: an entire generation did that and was lost. There are known risks, and risks that are as yet unknown.

It was a multifaceted look at new therapies and therapies yet to come, to prepare ourselves with knowledge, when the time comes to make an informed decision.
See photo gallery of the day here.
Kudos to NEHA for a wonderful day and event! Thanks to everyone who traveled so far to come and share their wisdom with us in Maine.

