1,691
Written by Leonard A. Valentino, MD
Originally published in YOU, August 2015
Steve is a lifelong Chicago White Sox fan and rarely misses a home game. For his birthday last year, a friend gave him a team ball cap to replace the one he’d worn for years. The new black and white cap looked cool and had a great brim for blocking the sun. But it was a non-adjustable, “one size fits all” model, and after just a couple of innings, Steve had to take it off because it was tight and uncomfortable. The cap was too large for his daughter but fit his son perfectly, and he became its new owner.
Standard prophylaxis is a lot like that baseball cap. For years, we’ve relied on a one size fits all approach to treatment. For many, these standard regimens, intended to keep FVIII or FIX trough levels above 1% of normal, prevent or reduce bleeding. Other patients may need higher trough levels to prevent bleeds, while some may be overtreated with a standard approach. So, how can we improve the “fit” of prophylaxis and reduce the risk of bleeding or overtreatment for everyone? Make the regimen adjustable by customizing treatment to the individual. In other words, personalizing prophylaxis.
Step One: Assessing Bleeding Risk
To personalize a prophylaxis regimen, your physician must first assess your risk for bleeding.1 This risk is partly determined by your bleeding phenotype—how often you bleed when not on prophylaxis. While many people with severe hemophilia have frequent, spontaneous joint bleeds, some rarely experience bleeding episodes. Your bleeding phenotype helps determine the intensity of prophylaxis. If you have a severe phenotype, you may require a higher dose of factor concentrate and/or more frequent infusions to prevent bleeding.
The types of activities a person participates in may be another risk for bleeding. Although an active lifestyle is essential for maintaining overall health, vigorous physical activity may somewhat increase the risk for bleeding in persons with hemophilia. Personalizing prophylaxis by raising FVIII or FIX levels during certain physical activities may help prevent or reduce injury-related (traumatic) bleeds.
Age at the start of prophylaxis and the presence of a target joint also contribute to bleeding risk. Hemophilia treaters continue to debate the ideal age for beginning prophylaxis. Yet they all agree that it is not a good idea to delay prophylaxis until someone experiences several bleeds— particularly, multiple bleeds into the same joint (a target joint). Once this situation develops, personalized prophylaxis may be needed to reduce a pattern of frequent bleeding.
Finally, FVIII or FIX pharmacokinetics (PK) has a major impact on bleeding risk. PK is a scientific approach to study and describe what the body does to a drug—in this case, clotting factor concentrate. Because this topic is so important, let’s consider it separately.
Your Pharmacokinetic Profile
One of the best tools we have for personalizing prophylaxis is a person’s unique PK profile. This profile, derived from blood samples taken before and after factor infusion, provides information about how your body uses factor concentrate. The main determinants of PK are how often you infuse and how long FVIII or FIX remains active afterwards.1
Following an infusion, factor level increases, with the increase determined by the amount of factor administered and the patient’s response to the infused dose. This information is used to calculate the incremental recovery. For example, a FVIII injection of 50 IU/kg body weight would be expected to increase the FVIII level to 100% of normal values, indicating an incremental recovery of 2 IU/dL per IU/kg infused. This level quickly begins to fall, however, and after about 12 hours for FVIII and 24 hours for FIX, the level drops to 50%.This decline is called factor half-life, meaning that your body has used up half of the infused factor.
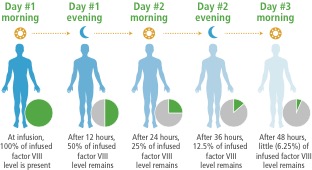 |
|
Figure 1. Factor VIII Half-Life Levels
|
 |
| Figure 2. Factor IX Half-Life Levels |
Although incremental recovery is fairly consistent among persons with hemophilia, factor half-life varies widely. In fact, a clinical study of prophylaxis found that some people have a half-life that is more than 2 times longer than that of someone with the shortest half-life. Personalizing prophylaxis for someone with a short factor half-life may involve infusing more often. Persons who are physically active may also benefit from personalizing prophylactic infusions. This strategy has worked well for several of my former patients.
On the other hand, persons with longer than average half-lives and those who are inactive because of severe joint disease or a sedentary lifestyle may be able to infuse less frequently. Ed* is a retired office worker with hemophilia A who has arthritis in both knees and uses a walker for mobility. He started prophylaxis at age 57 to reduce frequent knee bleeds. Although this regimen was effective, thrice-weekly infusions were a struggle for him, and he often missed doses. We measured his personal half-life and found he was at the high end of the normal range. We leveraged this long half-life and modified his FVIII prophylaxis regimen to every third day infusions. Since personalizing prophylaxis a year ago, Ed’s annual bleed rate has decreased from 24 to 30 to just 2.
Optimizing Success
An essential component of any personalized prophylaxis regimen is good adherence.1
Prophylaxis is unquestionably time-consuming and often inconvenient. However, bleeding resulting from missed doses is even more burdensome and can have negative consequences.
Here are a few suggestions for optimizing your success:
- Become an educated healthcare consumer by learning about your bleeding disorder and current treatment regimen. Consulting your local hemophilia chapter may be an important first step.
- Record all infusions in an electronic or paper bleed log and share this information with your healthcare providers.
Discuss your personal goals and aspirations with your healthcare team, making sure to emphasize how you define a successful treatment regimen. - If you have questions about any aspect of the treatment plan, contact your hemophilia treatment center (HTC) and speak with a nurse or doctor.
- If you experience breakthrough bleeding, immediately contact your HTC, as this may indicate a need to modify the prophylaxis regimen.
- When traveling, make certain to bring extra factor concentrate/infusion supplies in case of flight delays, etc.
Summing up, one size fits all doesn’t necessarily work for baseball caps or prophylaxis. Personalizing the prophylaxis regimen by taking into consideration your individual risk for bleeding and PK profile is key to ensuring a “good fit” that reduces bleeding while avoiding overtreatment. Once you and your physician have personalized your treatment plan, always infuse on schedule and immediately report any breakthrough bleeding. A little fine-tuning of the regimen to account for changes in your lifestyle may be all that is needed to prevent bleeds.
Dr. Valentino is a professor of pediatrics at Rush University in Chicago, Illinois, and the former director of the Rush Hemophilia and Thrombophilia Center. He is now the global head of Hemophilia & Blood Disorders, Global Medical Affairs at Shire.
Interested in more content like this?
Click Here to subscribe to our Parent Empowerment Newsletter.
Free to families with hemophilia
Free to families with hemophilia
* Names and some details have been changed to protect patient confidentiality.
1. Valentino, L. A. “Considerations in Individualizing Prophylaxis in Patients with Haemophilia A.” Haemaphilia 20(5) (2014): 607–15.

